Vertebroplasty
 Dr William Clark has pioneered the use of vertebroplasty in Australia for osteoporotic spinal fractures. He has performed more than 2,000 vertebroplasties over the last 20 years.
Dr William Clark has pioneered the use of vertebroplasty in Australia for osteoporotic spinal fractures. He has performed more than 2,000 vertebroplasties over the last 20 years.
Vertebroplasty is a treatment for painful vertebral fractures which occur in abnormally weak bone due to osteoporosis. Osteoporosis is common in patients over 60. It results in progressive loss of bone substance making the bones fragile and easily fractured. Fractures can be caused by a fall, recurrent stress on the bones from continual coughing or bending, from certain activities such as gardening, bowling, golf, lifting a heavy weight or even without trigger if the osteoporosis is severe.
Most osteoporotic spinal fractures cause mild symptoms, but some patients develop severe pain which makes normal every-day function and mobility difficult – particularly within the first 6-weeks of the fracture. In these patients with early fractures and severe pain, vertebroplasty can provide great relief. The side-effects of opiate pain killing medications such as constipation and confusion can be a problem. Vertebroplasty can be effective in reducing back pain and breaking the requirement on opiate medications.
Vertebroplasty is not useful for older, healed fractures or for chronic (long-standing) back pain. It is reserved for elderly patients with severe pain due to fracture of less than 6-weeks duration, who have severe pain.
Anatomy
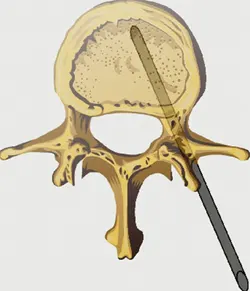
The vertebral body is a square shaped bone which bears the weight of the head, chest, and upper limbs. It is composed of an outer shell of “cortical” bone which surrounds the spongy or “trabecular” bone which constitutes the majority of the vertebral body. It is the trabecular bone which fractures. Vertebroplasty uses a needle to inject bone cement into the trabecular bone in the centre of the vertebral body. The shell of cortical bone serves to constrain the cement within the fractured trabecular bone of the vertebral body. The cement sets hard within 10 minutes and restores integrity to the bone. The cement distributes form top to bottom and side to side of the vertebral body and once again makes it a functional building block, as it was prior to the fracture. When the patient gets out of bed and stands up, rather than squashing down under the weight of the upper torso, the fractured bone is now resilient and able to bear the wight of the body above without squashing. This causes substantial pain relief – with the ghastly pain of fracture instability no longer present. The patient will regain mobility and return to normal function more quickly.
How is the procedure performed?
Vertebroplasty is performed using intravenous sedation to render the patient relaxed and sleepy. The patient is lying in the prone position (on the stomach) with an oxygen mask and with an oxygen finger monitor to ensure that the oxygen levels in the blood are always normal. Local anaesthetic is administered to the area of injection and then a needle is passed into the vertebral bone using imaging guidance. The imaging machine is an interventional fluoroscope which provides real time imaging of the needle movement and the vertebral bones on a TV screen. When the needle is in good position, the cement is injected slowly and gradually fills the fracture cleft and trabecular bone inside the vertebral body. This is watched in real time on the screen which makes the procedure safe. The whole procedure for one fracture takes about 30 minutes. For additional fractures extra time may be needed.
The cement used is high viscosity polymethyl-methacrylate (PMMA). PMMA has been used in other orthopaedic applications for more than 40 years and is known to be safe. It is slowly injected into the bone under real time imaging as the doctor watches it distribute in the vertebral body. When the distribution of cement is adequate to fully support the fractured bone, the injection is ceased.
What are the risks?
The risks of vertebroplasty are low when the operator is experienced and using high quality imaging equipment. Cement can potentially leak outside of the vertebral body during injection. The interventional radiologist monitors the spread of cement on the imaging screen in real time and If this begins to occur the injection is ceased. This provides the safety factor. The main risk is infection which is rare – occurring in about one per thousand cases.
History of Medicare funding
The role for vertebroplasty in the management of osteoporotic spinal fractures has been controversial. Vertebroplasty has been widely practiced in the USA, Europe, and Australia for more than 20 years. The procedure was previously funded by Medicare and by private health funds until 2011, when Medicare funding was terminated. Because the health insurance funds only pay for procedures which are listed on the Medicare schedule, they also ceased payment for vertebroplasty.
The Medicare Services Advisory Committee (MSAC), which advises the Government about Medicare funding, recommended termination of Medicare funding for vertebroplasty in 2011 because two studies showed no benefit from vertebroplasty. One of these was a small Australian study completed in Melbourne and the other was a larger international trial in which our practice had participated. These were chronic fracture trials, which studied the use of vertebroplasty in patients with fractures up to 12 months old. We advised MSAC at the time, that although the trials were useful in proving that vertebroplasty was not effective in treating healed fractures, that these two trials were not relevant to the practice of vertebroplasty at St George Private Hospital in Sydney – where we treat fresh fractures less than 6-weeks old in patients with severe pain.
VAPOUR Trial
To gather evidence for vertebroplasty in this group of patients, with fresh fractures and severe pain, we conducted the VAPOUR trial which was published in the prestigious Lancet journal in 2016. The VAPOUR trial was conducted in four hospitals in Sydney. Dr William Clark was the lead investigator. Between 2011 and 2014 we recruited 120 patients with fractures less than 6-weeks duration who had severe pain. We recorded pain, disability, and medication usage data at 3-days, 2-weeks, 1 month, 3-months and 6 months after the procedure. Half the patients had vertebroplasty and the other half had a “placebo” procedure, where we pretended to do a vertebroplasty. The patients did not know which procedure they had undergone – so they were “blinded” as to the procedure. This kind of placebo or “blinded” trial provides the highest level of evidence that a trial can obtain. The VAPOUR trial proved that vertebroplasty resulted in better pain reduction than the placebo at every time point from 3-days to 6-months after the procedure. Vertebroplasty also provided faster reduction in disability, reduced requirement of pain medications and reduced the requirement of hospitalisation in the vertebroplasty group compared to the placebo group.
Return of Medicare funding in 2021
Based on this new evidence from the VAPOUR trial, the MSAC has recommended resumption of Medicare funding for vertebroplasty for patients with new fractures affecting the thoracolumbar segment of the spine. This decision was published on the MSAC site in April 2020, but the Department of Health has been slow to act on the recommendation, blaming the coronavirus for the delay. It is scheduled for passage through the Australian Parliament in May 2021, which should see the Medicare and Private health funding for vertebroplasty be recommenced by July 2021.
The VAPOUR trial is attached on this web-site for those who are interested to read it.